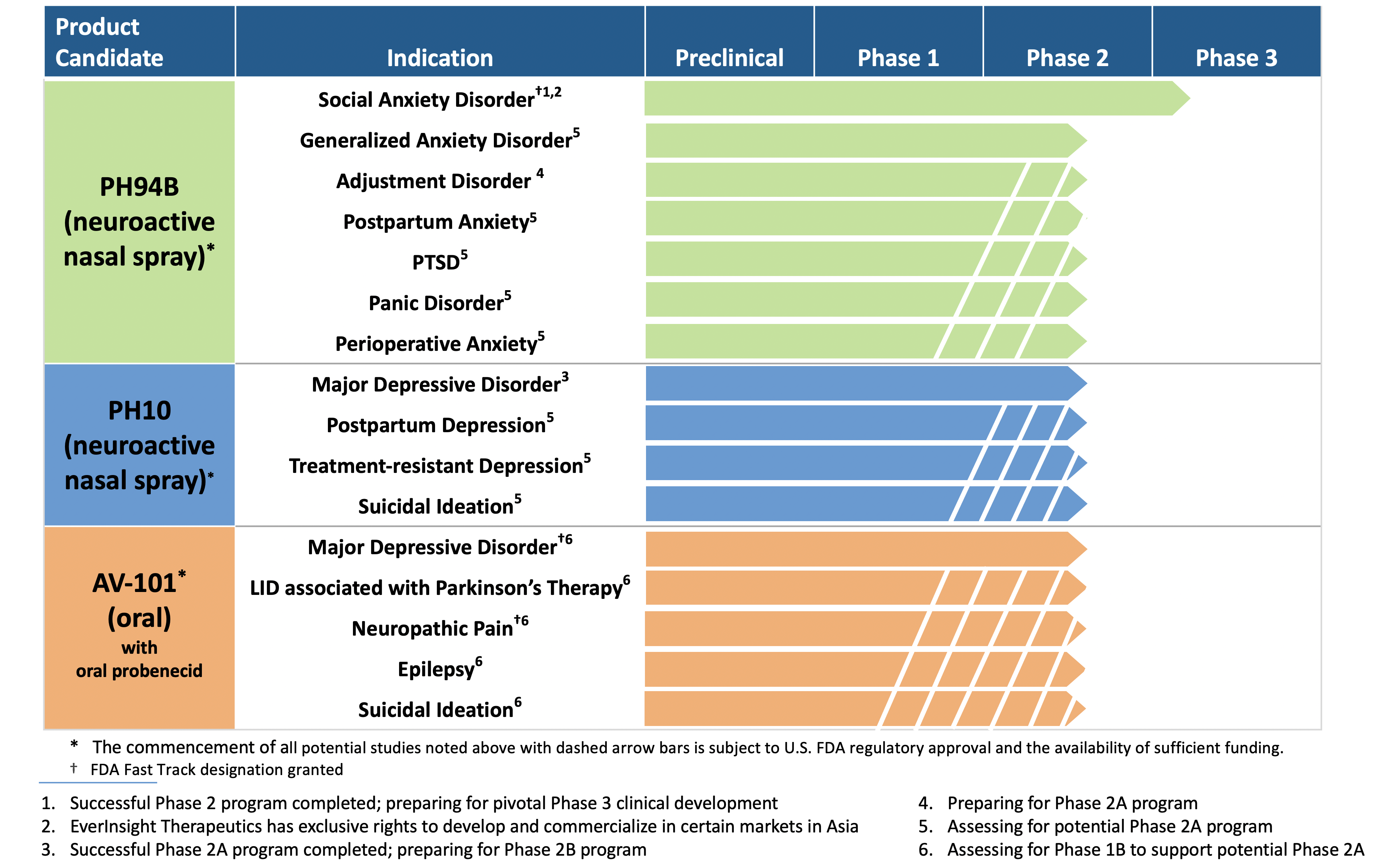
VistaGen says FDA found no signal of abuse potential for its nasal spray PH94B for anxiety | Seeking Alpha

VistaGen readies Phase 3 trial of novel, fast-acting neuroactive nasal spray in social anxiety disorder - BioTuesdays

VistaGen Reports Positive Preclinical Data Differentiating Mechanism of Action of PH94B from Risk-Ridden Benzodiazepines

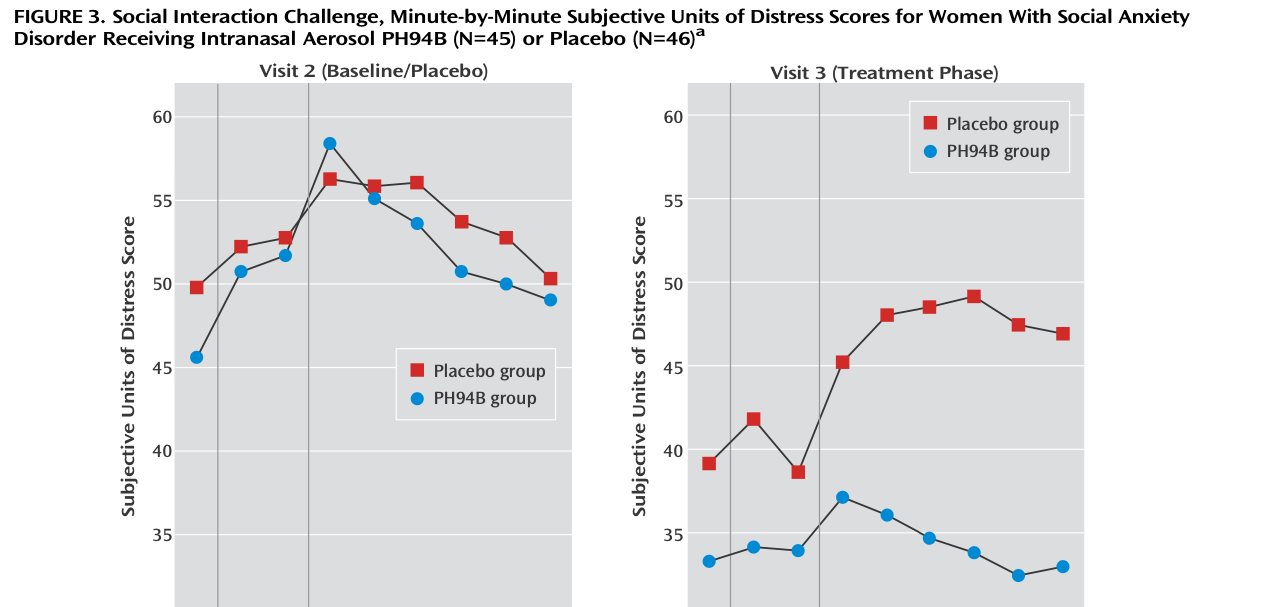
PH94B Neuroactive Nasal Spray, a Potential On-Demand Treatment for Social Anxiety Disorder | Learn how PH94B, a novel, rapid-acting CNS neuroactive nasal spray being developed by VistaGen Therapeutics for the treatment of

PH94B Neuroactive Nasal Spray, a Potential On-Demand Treatment for Social Anxiety Disorder - YouTube

VistaGen readies Phase 3 trial of novel, fast-acting neuroactive nasal spray in social anxiety disorder - BioTuesdays

U.S. FDA Grants VistaGen Therapeutics Fast Track Designation for PH94B for Treatment of Social Anxiety Disorder

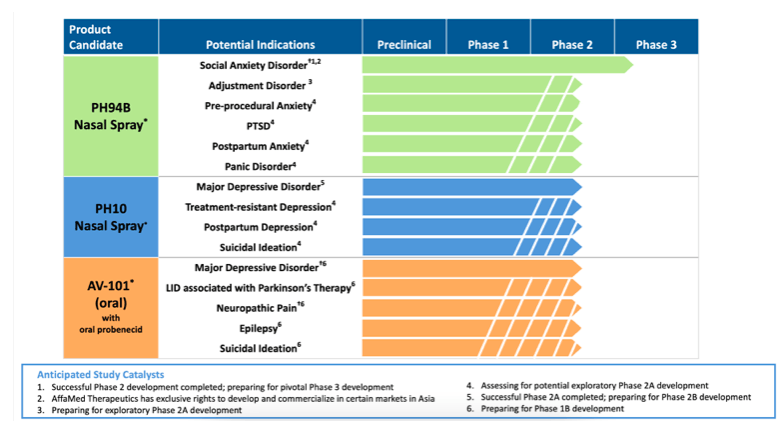
VistaGen to Present PH94B Exploratory Phase 2A Research Program for Adjustment Disorder with Anxiety at American Society for Clinical Psychopharmacology Annual Meeting






,%20Phobia,%20Social,%20Anxiety%20Disorders:%0A%0APH94B%20Nasal%20Spray%20for%20Social%20Anxiety%20Disorder%20(SAD).png?md=1)

:%0A%0APH94B%20Nasal%20Spray%20for%20Anxiety%20Disorders.png?md=1)